Chemistry, 23.11.2019 03:31 evelynnn452
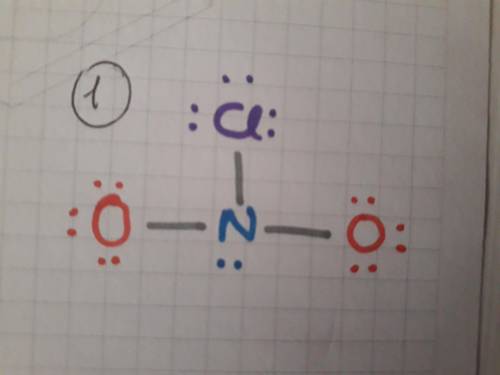
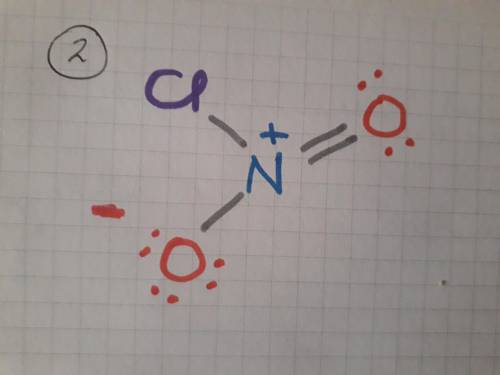
Draw the lewis structure for each of the following and then determine if the molecule is polar or nonpolar. do not draw double bonds to oxygen atoms unless they are needed for the central atom to obey the octet rule. do not include formal charges in your drawing. no2cl.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:30
How much energy moves onto the next level, in an energy pyramid
Answers: 1

Chemistry, 21.06.2019 19:30
Water molecules have a strong attraction to each other because of hydrogen bonding, allowing water to move against gravity up a plant's stem through capillary action. true false
Answers: 2

Chemistry, 21.06.2019 23:40
If the atomic mass of an atom is 34 and the atom contains 13 protons, how many neutrons does the atom contain?
Answers: 2

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1
You know the right answer?
Draw the lewis structure for each of the following and then determine if the molecule is polar or no...
Questions




History, 07.12.2020 17:30


Computers and Technology, 07.12.2020 17:30


English, 07.12.2020 17:30



Engineering, 07.12.2020 17:30



English, 07.12.2020 17:30