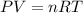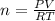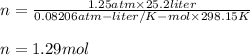Chemistry, 24.11.2019 05:31 aliyyahlove
you have a 25.2 l sample of gas at 1.25 atm and 25.0 degrees celsius. how many moles are present in this gas. for your answer, do not include units and provide your answer to three significant figures.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:00
What is the result of multiplying (2.5 × 1010) × (2.0 × 10-7)? a. 5.0 × 103 b. 5.0 × 10-3 c. 5.0 × 1017 d. 5.0 × 10-17
Answers: 1

Chemistry, 22.06.2019 06:00
An atom of lithium (li) and an atom of chlorine (cl) engage in a chemical reaction. which correctly describes the structure of the resulting chemical compound? hint: consider the class of each element. the chemical compound will have a network structure. the chemical compound will have triple bonds. the chemical compound will have a ball-and-stick structure. the chemical compound will have double bonds.
Answers: 2

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1
You know the right answer?
you have a 25.2 l sample of gas at 1.25 atm and 25.0 degrees celsius. how many moles are present in...
Questions






Mathematics, 02.07.2020 21:01



Mathematics, 02.07.2020 21:01

Mathematics, 02.07.2020 21:01

Mathematics, 02.07.2020 21:01



Mathematics, 02.07.2020 21:01

Mathematics, 02.07.2020 21:01



Mathematics, 02.07.2020 21:01


Biology, 02.07.2020 21:01