Chemistry, 25.11.2019 19:31 klocke2001
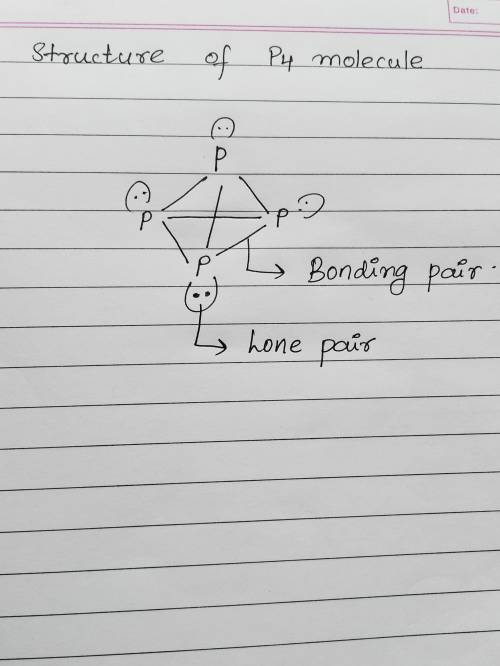
White phosphorus is composed of tetrahedral molecules of p4 in which every p atom is connected to three other p atoms. in the lewis structure of p4, there are 1. 6 bonding pairs and 4 lone pairs of electrons. 2. 6 bonding pairs and no lone pairs of electrons. 3. 5 bonding pairs and 4 lone pairs of electrons. 4. 3 bonding pairs and 4 lone pairs of electrons. 5. 6 bonding pairs and 2 lone pairs of electrons.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Which of the following did jj thompson discover about atoms? a)an atom has an internal structure. b) atoms are tiny indivisible particles. c)electrons orbit the nucleus of an atom. d) the nucleus of an atom contains protons and neutrons.
Answers: 2

Chemistry, 22.06.2019 12:10
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3

Chemistry, 22.06.2019 16:00
How will the volume of a gas be affected if the pressure is tripled, but the temperature remains the same?
Answers: 3

Chemistry, 22.06.2019 20:00
Glucose (c6h12o6) is an important biological molecule. (round the answer to nearest hundredth.) what is the percent by mass of carbon in glucose?
Answers: 2
You know the right answer?
White phosphorus is composed of tetrahedral molecules of p4 in which every p atom is connected to th...
Questions




Physics, 11.07.2019 15:00


Mathematics, 11.07.2019 15:10

Mathematics, 11.07.2019 15:10


Physics, 11.07.2019 15:10

Biology, 11.07.2019 15:10


Biology, 11.07.2019 15:10


Mathematics, 11.07.2019 15:10


Mathematics, 11.07.2019 15:10