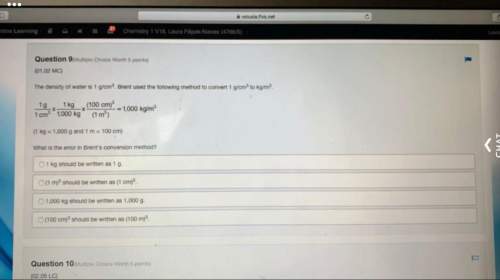
Aclosed vessel system of volume 2.5 l contains a mixture of neon and fluorine. the total pressure is 3.32 atm at 0.0°c. when the mixture is heated to 15°c, the entropy of the mixture increases by 0.345 j/k. what amount (in moles) of each substance (ne and f2) is present in the mixture? (heat capacity of monoatomic gas = 3/2r and that for a diatomic gas = 5/2r)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:30
What type(s) of intermolecular forces are expected between ch3ch2cooh molecules? dipole forces, induced dipole forces, hydrogen bonding
Answers: 1

Chemistry, 22.06.2019 21:30
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1

Chemistry, 23.06.2019 00:00
#20 which type of bond is formed when bases pair in dna? ionic bond covalent bond coordinate bond hydrogen bond
Answers: 1

Chemistry, 23.06.2019 01:20
How can parts of a solution be separated by chromatography?
Answers: 1
You know the right answer?
Aclosed vessel system of volume 2.5 l contains a mixture of neon and fluorine. the total pressure is...
Questions

Medicine, 06.08.2019 04:10

Geography, 06.08.2019 04:10


Mathematics, 06.08.2019 04:10


English, 06.08.2019 04:10

Mathematics, 06.08.2019 04:10

Biology, 06.08.2019 04:10

Chemistry, 06.08.2019 04:10