Answer : The molal freezing point depression constant of X is 
Explanation : Given,
Mass of urea (solute) = 5.90 g
Mass of X liquid (solvent) = 450.0 g
Molar mass of urea = 60 g/mole
Formula used :
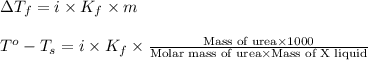
where,
 = change in freezing point
= change in freezing point
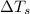 = freezing point of solution =
= freezing point of solution = 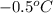
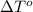 = freezing point of liquid X=
= freezing point of liquid X= 
i = Van't Hoff factor = 1 (for non-electrolyte)
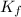 = molal freezing point depression constant of X = ?
= molal freezing point depression constant of X = ?
m = molality
Now put all the given values in this formula, we get
![[0.4-(-0.5)]^oC=1\times k_f\times \frac{5.90g\times 1000}{60g/mol\times 450.0g}](/tpl/images/0390/6513/c28e3.png)

Therefore, the molal freezing point depression constant of X is 


























