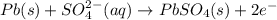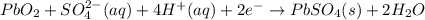Chemistry, 26.11.2019 03:31 giannimaraj711
In a lead-acid battery, the electrodes are consumed. in this battery, select one: a. the anode is pbo2. b. the cathode is pb. c. the cathode is pbso4. d. the anode is pbso4. e. the anode is pb.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:30
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 21.06.2019 19:30
If the element whose electric configuration ends in the d sublevel, the element is calssified as? a.inner transition b.noble gases c.representative d. transition
Answers: 2

Chemistry, 22.06.2019 11:40
Enzymes affect the reactions in living cells by changing the
Answers: 3

Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2
You know the right answer?
In a lead-acid battery, the electrodes are consumed. in this battery, select one: a. the anode is p...
Questions


Mathematics, 03.02.2021 14:00

English, 03.02.2021 14:00


Advanced Placement (AP), 03.02.2021 14:00

Mathematics, 03.02.2021 14:00


Medicine, 03.02.2021 14:00


History, 03.02.2021 14:00





Mathematics, 03.02.2021 14:00




Mathematics, 03.02.2021 14:00

English, 03.02.2021 14:00