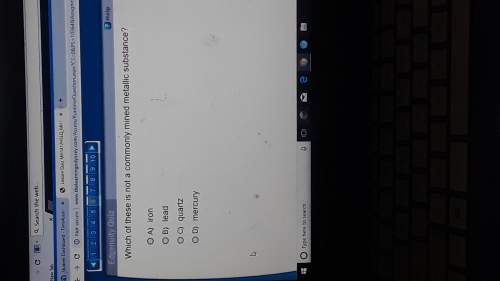
Chemistry, 26.11.2019 07:31 darriennichole
Consider the reactions _1 2 n2(g) + _1 2 o2(g) → no(g) _1 2 n2(g) + o2(g) → no2(g) if these reactions come to equilibrium after combustion in an internal-combustion engine at 2000 k and 200 bar, estimate the mole fractions of no and no2 present for mole fractions of nitrogen and oxygen in the combustion products of 0.70 and 0.05.

Answers: 3


Another question on Chemistry


Chemistry, 21.06.2019 22:30
How many moles are in 250 grams of tungsten (w)? * 4.4x10^23 moles 4.2x10^23 moles 0.7 moles 1.4 moles
Answers: 3


Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 3
You know the right answer?
Consider the reactions _1 2 n2(g) + _1 2 o2(g) → no(g) _1 2 n2(g) + o2(g) → no2(g) if these reaction...
Questions




Mathematics, 18.06.2020 00:57

English, 18.06.2020 00:57


Mathematics, 18.06.2020 00:57

Physics, 18.06.2020 00:57


Mathematics, 18.06.2020 00:57

Mathematics, 18.06.2020 00:57

Mathematics, 18.06.2020 00:57