Chemistry, 26.11.2019 21:31 Ciarrathereal
Achemist measures the energy change δh during the following reaction: c3h8 (g) +5o2 (g) →3co2 (g) +4h2o (l) =δh−2220.kj use the information to answer the following questions.
this reaction
endothermic.
exothermic.
suppose
81.0g
of
c3h8
react.
will any heat be released or absorbed?
yes, absorbed.
yes, released.
no.
if you said heat will be released or absorbed in the second part of this question, calculate how much heat will be released or absorbed.
kj
round your answer to
3
significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:30
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1

Chemistry, 22.06.2019 09:00
Ineed to find the answer of this question because i dont understand it
Answers: 1

Chemistry, 22.06.2019 21:20
One way in which the useful metal copper is produced is by dissolving the mineral azurite, which contains copper(ii) carbonate, in concentrated sulfuric acid. the sulfuric acid reacts with the copper(ii) carbonate to produce a blue solution of copper(ii) sulfate. scrap iron is then added to this solution, and pure copper metal precipitates out because of the following chemical reaction: (s) (aq) (s) (aq) suppose an industrial quality-control chemist analyzes a sample from a copper processing plant in the following way. he adds powdered iron to a copper(ii) sulfate sample from the plant until no more copper will precipitate. he then washes, dries, and weighs the precipitate, and finds that it has a mass of .
Answers: 2

Chemistry, 23.06.2019 00:00
What is the approximate mass of 25 cm3 of silver, if the density is 10.5 g/cm3? a. 0.42 g b. 2.4 g c. 42 g d. 260 g
Answers: 1
You know the right answer?
Achemist measures the energy change δh during the following reaction: c3h8 (g) +5o2 (g) →3co2 (g) +...
Questions


Mathematics, 05.09.2020 06:01



Mathematics, 05.09.2020 06:01






Social Studies, 05.09.2020 06:01


Mathematics, 05.09.2020 06:01

Mathematics, 05.09.2020 06:01



Biology, 05.09.2020 06:01


Mathematics, 05.09.2020 06:01

Mathematics, 05.09.2020 06:01

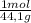 ×
×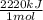 = 4,08x10³ kJ
= 4,08x10³ kJ