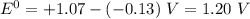Chemistry, 29.11.2019 00:31 savyblue1724707
Use the standard half-cell potentials listed below to calculate the standard cell potential for the following reaction occurring in an electrochemical cell at 25°c. (the equation is balanced.) pb(s) + br2(l) → pb2+(aq) + 2br(aq) pb2+(aq) + 2 e → pb(s) e° = -0.13 v br2(l) + 2 e → 2 br(aq) e° = +1.07 v

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 21:00
In the experiment you asked to react hydrochloric acid and with sodium hydroxide. when measuring the volume of the reactants, which instrument would give the greatest precision.
Answers: 3

Chemistry, 22.06.2019 23:00
Movement that is like a t a type of wave that transfers energy where the particles in the medium move in a circle motion while the energy travels left or right. a type of wave that transfers energy where the particles in the medium move perpendicular to the direction in which the energy is traveling. transfers energy from one location to another a type of wave that transfers energy where the particles in the medium move parallel to the direction in which the energy is traveling. movement that is back and forth, like an equal sign = 1. wave 2. parallel movement 3. perpendicular movement 4. transverse wave 5. longitudinal wave 6. surface wave
Answers: 1

Chemistry, 23.06.2019 02:40
Calculate the standard enthalpy of formation of liquid methanol, ch3oh(l), using the following information: c(graphite) + o2 latex: \longrightarrow ⟶ co2(g) latex: \delta δ h° = –393.5 kj/mol h2(g) + o2 latex: \longrightarrow ⟶ h2o(l) latex: \delta δ h° = –285.8 kj/mol ch3oh(l) + o2(g) latex: \longrightarrow ⟶ co2(g) + 2h2o(l) latex: \delta δ h° = –726.4 kj/mol
Answers: 3
You know the right answer?
Use the standard half-cell potentials listed below to calculate the standard cell potential for the...
Questions


History, 26.07.2019 21:00



Mathematics, 26.07.2019 21:00


Biology, 26.07.2019 21:00













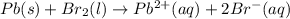
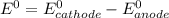
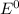 are standard reduction potentials.
are standard reduction potentials.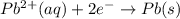
![E^0_{[Pb^{2+}/Pb]}= -0.13\ V](/tpl/images/0395/3322/82712.png)
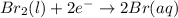
![E^0_{[Br_2/Br^{-}]}=+1.07\ V](/tpl/images/0395/3322/f8f7e.png)
![E^0=E^0_{[Br_2/Br^{-}]}- E^0_{[Pb^{2+}/Pb]}](/tpl/images/0395/3322/fba3a.png)