
Chemistry, 30.11.2019 07:31 sophiateaches053
When a 3.23 g sample of solid sodium hydroxide was dissolved in a calorimeter in 100.0 g of water, the temperature rose from 23.9 °c to 32.0 °c. calculate δh (in kj/mol naoh) for the following solution process: naoh(s) →na+(aq)+ oh−(aq)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
When curium-242 is bombarded with an alpha particle, two products are formed, one of which is a nudge on. what is the other product
Answers: 3

Chemistry, 22.06.2019 14:00
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3

Chemistry, 22.06.2019 22:30
What is a number added in front of a formula in order to balance the equation
Answers: 1

Chemistry, 23.06.2019 07:00
The image compares the arrangement of electrons in two different neutral atoms. a figure labeled atom q has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has six black spheres. to the left of this figure is another figure labeled atom p. atom p has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has seven black spheres. which of the following best explains the position of the two atoms in the periodic table?
Answers: 2
You know the right answer?
When a 3.23 g sample of solid sodium hydroxide was dissolved in a calorimeter in 100.0 g of water, t...
Questions





Computers and Technology, 26.12.2019 03:31







Mathematics, 26.12.2019 03:31


Biology, 26.12.2019 03:31






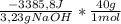 = -41929J/molNaOH ≡
= -41929J/molNaOH ≡ 

