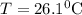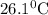Chemistry, 30.11.2019 07:31 Sylnatius6736
A25.5 −g aluminum block is warmed to 65.9 ∘c and plunged into an insulated beaker containing 55.2 g water initially at 22.1 ∘c. the aluminum and the water are allowed to come to thermal equilibrium.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 13:20
Determine which intermolecular forces are the dominant (strongest) forces for a pure sample of each of the following molecules by placing the molecules into the correct bins. drag the appropriate molecular formula to their respective bins.
Answers: 3


Chemistry, 21.06.2019 23:00
Achef makes salad dressing by mixing oil, vinegar, and spices, as shown. which type of matter is the salad dressing?
Answers: 1

Chemistry, 22.06.2019 00:30
Butadiene undergoes a reaction at a certain temperature in the gas phase as follows: 2c4h6(g) --> c8h12(g) the following data were collected for this reaction: time (min) [c4h6] (m) 0 0.36 15 0.30 30 0.25 48 0.19 75 0. determine the order of the reaction and the rate constant. 1st order and k = 4.3x10 -4 s-1 1st order and k = 2.3x10-4 s-1 2nd order and k = 4.3x10-4 s-1 2nd order and k = 2.3x10-4 s-1 zero and k = 4.3x10-4 s-1
Answers: 3
You know the right answer?
A25.5 −g aluminum block is warmed to 65.9 ∘c and plunged into an insulated beaker containing 55.2 g...
Questions

Mathematics, 25.11.2019 08:31

History, 25.11.2019 08:31

Mathematics, 25.11.2019 08:31

English, 25.11.2019 08:31

Medicine, 25.11.2019 08:31

History, 25.11.2019 08:31

Mathematics, 25.11.2019 08:31

Social Studies, 25.11.2019 08:31

English, 25.11.2019 08:31


Mathematics, 25.11.2019 08:31


History, 25.11.2019 08:31

Mathematics, 25.11.2019 08:31

Mathematics, 25.11.2019 08:31

Mathematics, 25.11.2019 08:31



Mathematics, 25.11.2019 08:31

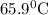 and plunged into an insulated beaker containing 55.2 g water initially at 22.1^{0}\textrm{C}. The aluminum and the water are allowed to come to thermal equilibrium. Assuming that no heat is lost, what is the final temperature of the water and aluminum?"
and plunged into an insulated beaker containing 55.2 g water initially at 22.1^{0}\textrm{C}. The aluminum and the water are allowed to come to thermal equilibrium. Assuming that no heat is lost, what is the final temperature of the water and aluminum?"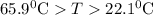
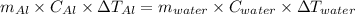
 represents change in temperature
represents change in temperature