
Chemistry, 05.12.2019 11:31 420420blazee
Ineed i don’t know if this’s right can you fill in the blanks
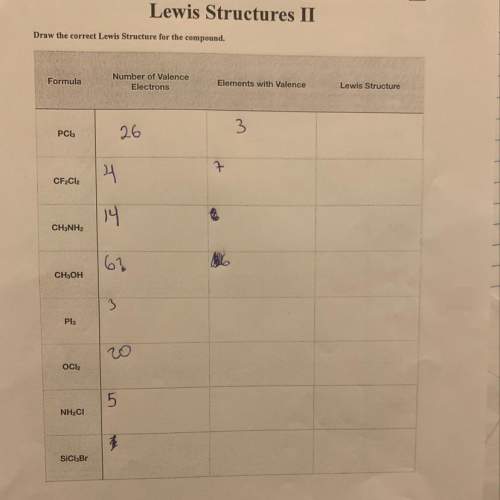

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 07:10
An experimental procedure requires a 10 ml of acid to be dissolved
Answers: 2

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1

Chemistry, 23.06.2019 00:30
The footprints of a dinosaur and the burrow of an ancient shrimp are examples of which kind of fossils
Answers: 2
You know the right answer?
Ineed i don’t know if this’s right can you fill in the blanks
...
...
Questions

Chemistry, 26.03.2020 18:14

Biology, 26.03.2020 18:14


Chemistry, 26.03.2020 18:14







Mathematics, 26.03.2020 18:14


Mathematics, 26.03.2020 18:14

Biology, 26.03.2020 18:14


Mathematics, 26.03.2020 18:14



Arts, 26.03.2020 18:14



