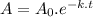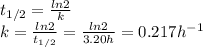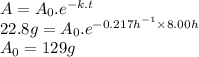Chemistry, 05.12.2019 21:31 aliviafrancois2000
An unknown radioactive substance has a half-life of 3.20 hours . if 22.8 g of the substance is currently present, what mass a0 was present 8.00 hours ago? express your answer with the appropriate units.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Which statements are true about electrolysis? check all that apply. electrolysis requires an acid be present. electrolysis is described by two half-reactions. electrolysis is not an industrial process. electrolysis results in commercially valuable products. electrolysis involves the transfer of electrons. reduction results in the loss of electrons. oxidation results in the loss of electrons.
Answers: 1

Chemistry, 22.06.2019 12:40
In the following table, all the columns for the element calcium are filled out correctly. element electron structure of atom electron structure of ion net ionic charge calcium 1s22s22p63s23p64s2 1s32s22p63s23p64s1 +1 true false
Answers: 2

Chemistry, 23.06.2019 09:00
How many grams of ammonia are produced when 1.0 mole of nitrogen reacts
Answers: 2

Chemistry, 23.06.2019 13:30
Why hydrochloric acid neutralized first when you titrate a mixture of hcl& ch3cooh against standard sodium hydroxide
Answers: 1
You know the right answer?
An unknown radioactive substance has a half-life of 3.20 hours . if 22.8 g of the substance is curre...
Questions





SAT, 18.03.2021 14:00

Physics, 18.03.2021 14:00

Mathematics, 18.03.2021 14:00


Mathematics, 18.03.2021 14:00


English, 18.03.2021 14:00

Business, 18.03.2021 14:00

Mathematics, 18.03.2021 14:00


Mathematics, 18.03.2021 14:00

Mathematics, 18.03.2021 14:00

Physics, 18.03.2021 14:00



Chemistry, 18.03.2021 14:00