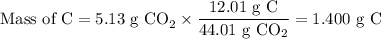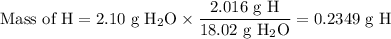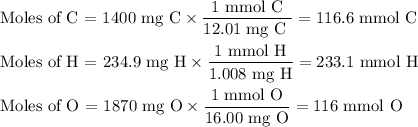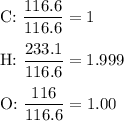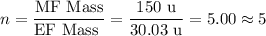Chemistry, 05.12.2019 23:31 aaleeyahprice
3.5g of a certain compound x, known to be made of carbon, hydrogen, and perhaps oxygen, and to have a molecular molar mass of 150g/mol is burned completely in excess oxygen, and the mass of the products carefully measured .
product
carbon dioxide 5.13g
water 2.10g
use this information to find the molecular formula of x

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Which of the following compounds does not contain molecules? question 2 options: co2 h2 nacl h2o
Answers: 1

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1

Chemistry, 22.06.2019 17:00
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2
You know the right answer?
3.5g of a certain compound x, known to be made of carbon, hydrogen, and perhaps oxygen, and to have...
Questions


Social Studies, 28.07.2021 01:00

History, 28.07.2021 01:00










Biology, 28.07.2021 01:00



Social Studies, 28.07.2021 01:00