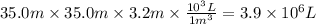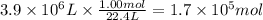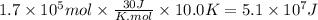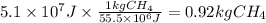Chemistry, 06.12.2019 17:31 keshewar2671
Assuming that all the energy given off in the reaction goes to heating up only the air in the house, determine the mass of methane required to heat the air in a house by 10.0 ∘c. assume each of the following: house dimensions are 35.0 m × 35.0 m × 3.2 m ; specific heat capacity of air is 30 j/k⋅mol; 1.00 mol of air occupies 22.4l for all temperatures concerned.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3

Chemistry, 22.06.2019 15:00
According to the diagram, what sources contribute to the phosphorus found in soil? according to the diagram, phosphorus found in soil contributes phosphorus to what other sources?
Answers: 1

Chemistry, 22.06.2019 18:30
What volume of a 0.0606 m solution of strontium bromide is needed to obtain 0.340 mol of the compound? question 42 options: a)5.61 l b) 3.4 l c) 600 ml d) 1 l e) 178 ml
Answers: 1

Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1
You know the right answer?
Assuming that all the energy given off in the reaction goes to heating up only the air in the house,...
Questions

Mathematics, 30.05.2021 22:20


English, 30.05.2021 22:20



English, 30.05.2021 22:30

Biology, 30.05.2021 22:30

Mathematics, 30.05.2021 22:30


Spanish, 30.05.2021 22:30


Chemistry, 30.05.2021 22:30


English, 30.05.2021 22:30


French, 30.05.2021 22:30




Mathematics, 30.05.2021 22:30