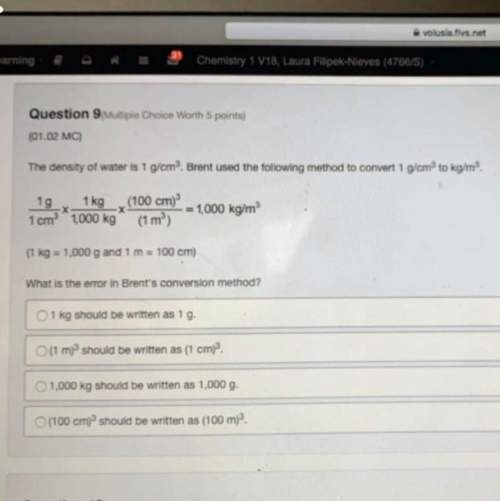
Based on your data, what is the likely identity (complete chemical formula) of the unknown sulfate? explain (include calculations as appropriate).
mass (g) of unknown sulfate = 0.53g
mass g of crucible= 28.63g
mass g of crucible + content= 29.44 g
mass of barium sulfate = .81g

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:40
In an effort to address concerns about global warming, a power plant in portland,oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

Chemistry, 22.06.2019 09:40
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3


Chemistry, 22.06.2019 11:20
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1
You know the right answer?
Based on your data, what is the likely identity (complete chemical formula) of the unknown sulfate?...
Questions




Mathematics, 25.05.2021 02:50

Mathematics, 25.05.2021 02:50




Chemistry, 25.05.2021 02:50






Mathematics, 25.05.2021 02:50



Mathematics, 25.05.2021 02:50