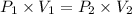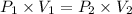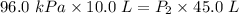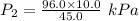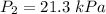Acylinder is filled with 10.0l of gas and a piston is put into it. the initial pressure of the gas is measured to be 96.0kpa. the piston is now pulled up, expanding the gas, until the gas has a final volume of 45.0l. calculate the final pressure of the gas. be sure your answer has the correct number of significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:30
A6.10 m nacl can be made by adding [x]g of nacl to a container and making the volume of water up to the 1.00 l line
Answers: 1


Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 06:30
The minerals found in bones are deposited by living cells called
Answers: 1
You know the right answer?
Acylinder is filled with 10.0l of gas and a piston is put into it. the initial pressure of the gas i...
Questions


Mathematics, 19.04.2021 20:40

Computers and Technology, 19.04.2021 20:40



Social Studies, 19.04.2021 20:40



Geography, 19.04.2021 20:40



Mathematics, 19.04.2021 20:40


Mathematics, 19.04.2021 20:40

Mathematics, 19.04.2021 20:40

History, 19.04.2021 20:40


Mathematics, 19.04.2021 20:40

History, 19.04.2021 20:40