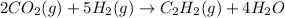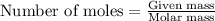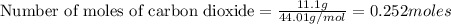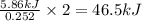Chemistry, 09.12.2019 20:31 poptropic9207
The reaction of carbon dioxide(g) with hydrogen(g) to form acetylene(g) (c2h2) and water(g) proceeds as follows: 2 co2(g) + 5 h2(g) c2h2(g) + 4 h2o(g) when 11.1 grams of co2(g) react with sufficient h2(g) , 5.86 kj of energy are absorbed . what is the value of h for the chemical equation given? δhrxn = kj

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 09:20
Explain that newton first law,second law and third law of motion?
Answers: 2


Chemistry, 22.06.2019 23:10
Amines are good nucleophiles, even though they are neutral molecules. how would the rate of an sn2 reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? amines are good nucleophiles, even though they are neutral molecules. how would the rate of an reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? because both reactants in the rate-limiting step are neutral, the reaction will be faster if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will be slower if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will occur at the same rate if the polarity of the solvent is increased. request answer
Answers: 3
You know the right answer?
The reaction of carbon dioxide(g) with hydrogen(g) to form acetylene(g) (c2h2) and water(g) proceeds...
Questions

Mathematics, 25.01.2022 02:00

Computers and Technology, 25.01.2022 02:00

English, 25.01.2022 02:00



English, 25.01.2022 02:00



Business, 25.01.2022 02:00


Mathematics, 25.01.2022 02:00


Mathematics, 25.01.2022 02:00

Physics, 25.01.2022 02:00

History, 25.01.2022 02:00


Arts, 25.01.2022 02:00

Mathematics, 25.01.2022 02:00

English, 25.01.2022 02:00