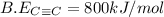Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:10
There are 6.022 x 10^23 atoms of hg in 1 mole of hg. the number of atoms in 4.5 moles of hg can be found by multiplying 4.5 by 6.022 x 10^23 a. 2.7 x 10^24 b. 27 x 10^23 c. 2.71 x10^24 d. 27.099 x 10^23
Answers: 3

Chemistry, 21.06.2019 23:00
The agent of mechanical weathering in which rock is worn away by the grinding action of other rock particles is call
Answers: 1

Chemistry, 22.06.2019 04:00
How do scientists think that gravity affected the formation of our solar system?
Answers: 1

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2
You know the right answer?
Acetylene gas (ethyne; hc≡ch) burns with oxygen in an oxyacetylene torch to produce carbon dioxide,...
Questions

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

English, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Geography, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

English, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

History, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

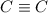 is 800 kJ/mol.
is 800 kJ/mol.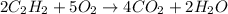
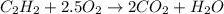
![\Delta H=[(B.E_{C\equiv C})+(2\times B.E_{C-H})+(2.5\times B.E_{O=O})]-[(2\times B.E_{C=O})+(2\times B.E_{H-O})]](/tpl/images/0410/6161/9cb32.png)
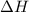 = heat of combustion = -1259 kJ/mol
= heat of combustion = -1259 kJ/mol = 413 kJ/mol
= 413 kJ/mol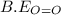 = 498 kJ/mol
= 498 kJ/mol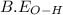 = 467 kJ/mol
= 467 kJ/mol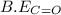 = 799 kJ/mol
= 799 kJ/mol![-1259kJ/mol=[(B.E_{C\equiv C})+(2\times 413kJ/mol)+(2.5\times 498kJ/mol)]-[(4\times 799kJ/mol)+(2\times 467kJ/mol)]](/tpl/images/0410/6161/6f294.png)