Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1

Chemistry, 22.06.2019 23:30
The density of the solid phase of a substance is 0.90 g/cm3 and the density of the liquid phase is 1.0 g/cm3. a large increase in pressure will a. lower the freezing point b. raise the freezing point c. lower the boiling point d. raise the triple point e. lower the triple point
Answers: 1

Chemistry, 23.06.2019 01:30
List and describe the neurological effects of the vocs and other air pollutants,as described by dr.theo colborn
Answers: 2

Chemistry, 23.06.2019 02:50
Dumbledore decides to gives a surprise demonstration. he starts with a hydrate of na2co3 which has a mass of 4.31 g before heating. after he heats it he finds the mass of the anhydrous compound is found to be 3.22 g. he asks everyone in class to determine the integer x in the hydrate: na2co3·xh2o; you should do this also. round your answer to the nearest integ
Answers: 2
You know the right answer?
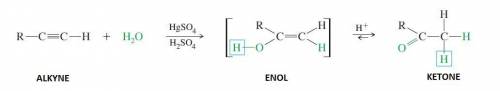
Identify the functional group that would be expected to be found in the final product upon completio...
Questions

Mathematics, 12.11.2020 22:10


History, 12.11.2020 22:10

Mathematics, 12.11.2020 22:10

Mathematics, 12.11.2020 22:10

Geography, 12.11.2020 22:10


Mathematics, 12.11.2020 22:10


Chemistry, 12.11.2020 22:10

Mathematics, 12.11.2020 22:10




Mathematics, 12.11.2020 22:10


Mathematics, 12.11.2020 22:10


Mathematics, 12.11.2020 22:10