
For the following reaction, 53.7 grams of iron(iii) oxide are allowed to react with 22.8 grams of aluminum. iron(iii) oxide (s) + aluminum (s) aluminum oxide (s) + iron (s) what is the maximum amount of aluminum oxide that can be formed? grams what is the formula for the limiting reagent? what amount of the excess reagent remains after the reaction is complete? grams

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:50
2points what is the job of a scientist? a. to answer ethical questions. b. to write laws based on his or her knowledge. c. to ask and answer scientific questions. d. to ignore facts that do not support his or her theory.
Answers: 1

Chemistry, 22.06.2019 00:00
Alarge marble is dropped in a graduated cylinder with 35ml of water in it.the water level increases to 49ml.what is the volume of the marble
Answers: 1

Chemistry, 22.06.2019 06:20
If i can still dissolve more sugar into the solution at a certain temperature what would i call that solution
Answers: 3

Chemistry, 22.06.2019 13:00
What happens to the average kinetic energy of a gas when the particles of the gas collide against each other at a constant temperature and volume? explain your answer.
Answers: 3
You know the right answer?
For the following reaction, 53.7 grams of iron(iii) oxide are allowed to react with 22.8 grams of al...
Questions



Physics, 14.12.2020 22:40

Health, 14.12.2020 22:40


Biology, 14.12.2020 22:40

Mathematics, 14.12.2020 22:40

Mathematics, 14.12.2020 22:40

Mathematics, 14.12.2020 22:40

Health, 14.12.2020 22:40


Health, 14.12.2020 22:40




Mathematics, 14.12.2020 22:40



History, 14.12.2020 22:40

Chemistry, 14.12.2020 22:40

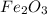 .
.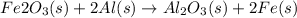
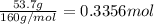
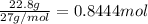
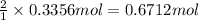 of aluminum.
of aluminum.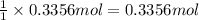 of aluminum oxide
of aluminum oxide

