
Chemistry, 11.12.2019 21:31 keniaguevara32
Calculate δhrxn for the following reaction: ch4(g)+2cl2(g)→ch2cl2(g)+2hcl(g) use the following reactions and given δh values. ch4(g)+cl2(g)→ch3cl(g)+hcl(g), δh=−99.60 kj ch3cl(g)+cl2(g)→ch2cl2(g)+hcl(g), δh=−105.8 kj express your answer to four significant figures.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:30
An empty fuel tank can still contain and therefore can be even more dangerous than one full of liquid fuel.
Answers: 1


Chemistry, 22.06.2019 03:00
Atrain travels 74 kilometers in 3 hours, and then 81 kilometers in 5 hours. what is its average speed?
Answers: 2

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1
You know the right answer?
Calculate δhrxn for the following reaction: ch4(g)+2cl2(g)→ch2cl2(g)+2hcl(g) use the following reac...
Questions

Mathematics, 30.04.2021 01:50



Mathematics, 30.04.2021 01:50

Mathematics, 30.04.2021 01:50



Mathematics, 30.04.2021 01:50

Mathematics, 30.04.2021 01:50

Mathematics, 30.04.2021 01:50



Chemistry, 30.04.2021 01:50

Mathematics, 30.04.2021 01:50

History, 30.04.2021 01:50

Mathematics, 30.04.2021 01:50



Mathematics, 30.04.2021 01:50

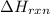 for the reaction is -205.4 kJ.
for the reaction is -205.4 kJ.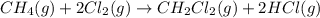
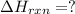
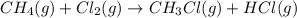
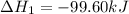
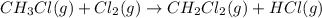
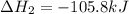
![\Delta H_{rxn}=[1\times \Delta H_1]+[1\times \Delta H_2]](/tpl/images/0414/0614/6e774.png)
![\Delta H_{rxn}=[(1\times (-99.60))+(1\times (-105.8))]=-205.4kJ](/tpl/images/0414/0614/3a575.png)


