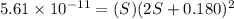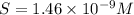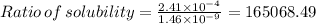Chemistry, 17.12.2019 02:31 winterblanco
Mg(oh)2 is a sparingly soluble salt with a solubility product, ksp, of 5.61×10−11. it is used to control the ph and provide nutrients in the biological (microbial) treatment of municipal wastewater streams. what is the ratio of solubility of mg(oh)2 dissolved in pure h2o to mg(oh)2 dissolved in a 0.180 m naoh solution? express your answer numerically as the ratio of molar solubility in h2o to the molar solubility in naoh.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:50
Which of the following electromagnetic waves can create ions?
Answers: 2

Chemistry, 23.06.2019 00:30
Ok, so i have 2 questions. try to answer them both: (the topic is fire) 1) how can you represent the chemical reaction of fire? 2) what kind of bond is formed in this chemical reaction
Answers: 3

Chemistry, 23.06.2019 04:00
If you are told to get 100 ml of stock solution to use to prepare smaller size sample for an experiment, which piece of glassware would you use?
Answers: 1

Chemistry, 23.06.2019 12:30
Idid a lab for chemistry where we put nails in a copper (ii) chloride solution. 1. why did the reaction stop? which reactant was used up? how do you know? 2. describe what was happening to the atoms of iron and copper during the reaction. what is this type of reaction called? 3. what would happen to the ratio of copper to iron if you had placed more nails in the beaker? if you had let the reaction go for less time? 4. what is the accepted ratio of copper atoms to iron atoms in this reaction? account for differences between your experimental value and the accepted value. write the balanced equation for the reaction.
Answers: 2
You know the right answer?
Mg(oh)2 is a sparingly soluble salt with a solubility product, ksp, of 5.61×10−11. it is used to con...
Questions


History, 03.07.2020 18:01


Mathematics, 03.07.2020 18:01














Mathematics, 03.07.2020 18:01

English, 03.07.2020 18:01

English, 03.07.2020 18:01

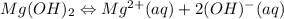
![[Mg^{2+}][OH^{-}]](/tpl/images/0421/6722/fd650.png)
![K_{sp}=[Mg^{2+}][OH^{-}]](/tpl/images/0421/6722/17c44.png)
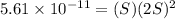
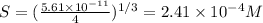
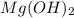 in 0.180 M NaOH is a follows.
in 0.180 M NaOH is a follows.