Carbon disulfide is prepared by heating sulfur and charcoal. the chemical equation is
 + c (s) \rightleftharpoons cs_2 (g)) ;
;  = 9.40 at 900 k
= 9.40 at 900 k
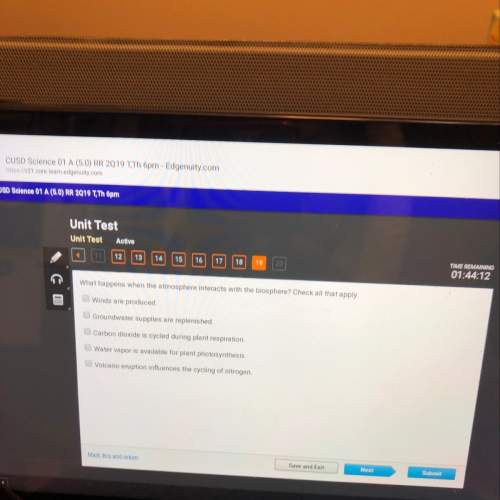
how many grams of cs₂ (g) can be prepared by heating 15.5 mol s₂ (g) with excess carbon in a 8.30 l reaction vessel held at 900 k until equilibrium is attained?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Oxidation-reduction reactions (often called "redox" for short) are reactions that involve the transfer of electrons from one species to another. oxidation states, or oxidation numbers, allow chemists to keep track of these electron transfers. in general, one element will lose electrons (oxidation), with the result that it will increase in oxidation number, and another element will gain electrons (reduction), thereby decreasing in oxidation number. the species that is oxidized is called the reducing agent or reductant. the species that is reduced is called the oxidizing agent or oxidant. to sum up: oxidation = increase in oxidation state = loss of electrons = reducing agent reduction = decrease in oxidation state = gain of electrons = oxidizing agent part a which element is oxidized in this reaction? fe2o3+3co→2fe+3co2 enter the elemental symbol. view available hint(s) is oxidized part b which element is reduced in this reaction? 2hcl+2kmno4+3h2c2o4→6co2+2mno2+2kcl+4h2o enter the elemental symbol. view available hint(s) is reduced
Answers: 1

Chemistry, 22.06.2019 06:00
How many atoms of mg are present in 97.22 grams of mg? 6.022 × 1023 2.408 × 1024 4.818 × 1024 5.855 × 1025
Answers: 3

Chemistry, 22.06.2019 07:30
Free answer. the treaty of versailles ended world war i, but some of the terms of the treaty contributed to the beginning of world war ii. which was one of the terms of the treaty? the answer would be "germany was forces to pay reparations to the allied countries.". i hope this .
Answers: 1

Chemistry, 22.06.2019 09:20
Sugar is dissolved in water. which is the solute? sugar neither both water
Answers: 1
You know the right answer?
Carbon disulfide is prepared by heating sulfur and charcoal. the chemical equation is
[tex]s_...
[tex]s_...
Questions

Mathematics, 09.10.2021 18:10



Mathematics, 09.10.2021 18:10


Social Studies, 09.10.2021 18:10

Business, 09.10.2021 18:10

Chemistry, 09.10.2021 18:10

Mathematics, 09.10.2021 18:10

History, 09.10.2021 18:10

SAT, 09.10.2021 18:10



English, 09.10.2021 18:10

Advanced Placement (AP), 09.10.2021 18:10

Mathematics, 09.10.2021 18:10




English, 09.10.2021 18:10

![Kc = \frac{[C]^c*[D]^d}{[A]^a*[B]*b}](/tpl/images/0422/1866/eff99.png)