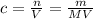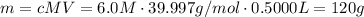Chemistry, 20.12.2019 21:31 ecoagro1934
Plss i'm really stuck!
how would you make a 500.0ml of stock solution of 6.0m of naoh. explain in detail with words and support with calculations.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:50
The name of the ion, s2-, is: sulfurous ion sulfide ion sulfur ion sulfate ion
Answers: 1

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 3

Chemistry, 22.06.2019 10:30
How do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 2
You know the right answer?
Plss i'm really stuck!
how would you make a 500.0ml of stock solution of 6.0m of naoh....
how would you make a 500.0ml of stock solution of 6.0m of naoh....
Questions



Law, 02.08.2021 04:20


English, 02.08.2021 04:20





Mathematics, 02.08.2021 04:20

Chemistry, 02.08.2021 04:20


Mathematics, 02.08.2021 04:20

Mathematics, 02.08.2021 04:20



Mathematics, 02.08.2021 04:20