
Chemistry, 24.12.2019 19:31 jeifetz1023
What is the theoretical yield of aluminum that can be produced by the reaction of 60.0 g of aluminum oxide with 30.0 g of carbon according to the following chemical equation? al2o3 + 3c → 2al + 3co

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:50
Someone offer some answers to this, i will give 98 coins and mark as brainliest! i will put the rest of the lab down in the comments,solutions pre-lab questions: in this lab, you will make fruit drinks with powdered drink mix. complete the pre-lab questions to get the values you need for your drink solutions. calculate the molar mass of powered fruit drink mix, made from sucrose (c12h22o11).using stoichiometry, determine the mass of powdered drink mix needed to make a 1.0 m solution of 100 ml. (hint: use molarity = to find the moles of drink mix, then convert moles to grams using a mole conversion.)what mass of powdered drink mix is needed to make a 0.5 m solution of 100 ml?
Answers: 1

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1

Chemistry, 23.06.2019 00:00
The graph indicates the running route for tobias. which best describes his run? from time 0 to 6, he went fast and then slowed down. from time 6 to 10, he was at his slowest. from time 12 to 14, he went very slow. from time 14 to 18, he went toward the starting point.
Answers: 2

Chemistry, 23.06.2019 02:30
Which of the four hypothetical substances you investigated would be most harmful to living organisms? 50 points!
Answers: 2
You know the right answer?
What is the theoretical yield of aluminum that can be produced by the reaction of 60.0 g of aluminum...
Questions




English, 08.03.2021 18:00

Social Studies, 08.03.2021 18:00

Mathematics, 08.03.2021 18:00




Mathematics, 08.03.2021 18:00

Mathematics, 08.03.2021 18:00

Mathematics, 08.03.2021 18:00


Mathematics, 08.03.2021 18:00






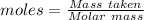
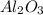
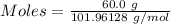
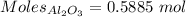
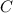
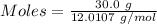
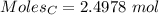
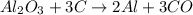
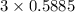 moles of carbon
moles of carbon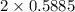 moles of aluminium.
moles of aluminium.

