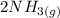Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Hot air balloons float in the air because of the difference in density between cold and hot air. in this problem, you will estimate the minimum temperature the gas inside the balloon needs to be, for it to take off. to do this, use the following variables and make these assumptions: the combined weight of the pilot basket together with that of the balloon fabric and other equipment is w. the volume of the hot air inside the balloon when it is inflated is v. the absolute temperature of the hot air at the bottom of the balloon is th (where th> tc). the absolute temperature of the cold air outside the balloon is tc and its density is ďc. the balloon is open at the bottom, so that the pressure inside and outside the balloon is the same. as always, treat air as an ideal gas. use g for the magnitude of the acceleration due to gravity.
Answers: 1

Chemistry, 22.06.2019 04:30
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1


Chemistry, 22.06.2019 12:30
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2
You know the right answer?
Hydrogen gas and nitrogen gas react to form ammonia gas. what volume of ammonia would be produced by...
Questions

Chemistry, 01.08.2019 19:30

Biology, 01.08.2019 19:30


Mathematics, 01.08.2019 19:30


Mathematics, 01.08.2019 19:30

Mathematics, 01.08.2019 19:30



Mathematics, 01.08.2019 19:30


Mathematics, 01.08.2019 19:30


Mathematics, 01.08.2019 19:30

Biology, 01.08.2019 19:30



Biology, 01.08.2019 19:30

Mathematics, 01.08.2019 19:30

Social Studies, 01.08.2019 19:30

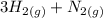 ⇒
⇒