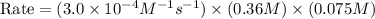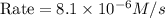Chemistry, 31.12.2019 00:31 texas101st78
The rate constant for the reaction nh4+(aq) + no2−(aq) --> n2(g) + 2h2o(l) is, k = 3.0 × 10^−4 m^-1 · s^-1. calculate the rate of the reaction if [nh4+] = 0.36 m and [no2−] = 0.075 m. (the reaction is in first order in regards to nh4+ as well as no2-) answer in scientific notation

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:30
Asample of the male sex hormone testosterone, c19h28o2, contains 3.88×10^21 atoms of hydrogen.(a) how many atoms of carbon does it contain? (b) how many molecules of testosterone does it contain? (c) how many moles of testosterone does it contain? (d) what is the mass of this sample in grams?
Answers: 1

Chemistry, 22.06.2019 04:00
Drag each label to the correct location on the chart. classify each reaction as endothermic or exothermic.
Answers: 1

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 23.06.2019 01:00
Which process results in the release of energy stored in the products of photosynthesis? a. polymer synthesis b. depolymerization c. digestion d. cellular respiration
Answers: 1
You know the right answer?
The rate constant for the reaction nh4+(aq) + no2−(aq) --> n2(g) + 2h2o(l) is, k = 3.0 × 10^−4 m...
Questions

History, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

History, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

History, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

Mathematics, 10.09.2020 19:01

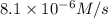
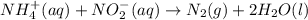
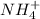 and
and 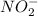 are the reactants.
are the reactants.![\text{Rate}=k[NH_4^+][NO_2^-]](/tpl/images/0437/6898/ed258.png)
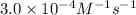
![[NH_4^+]](/tpl/images/0437/6898/5c46c.png) = concentration of
= concentration of ![[NO_2^-]](/tpl/images/0437/6898/10a69.png) = concentration of
= concentration of