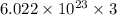Chemistry, 02.01.2020 04:31 lgisselle629
How many chlorine atoms would be in 6.02 x 10^23 units of gold iii chloride

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 09:00
Given the following reaction: c3h8+5o2=3co2+4h20 how many grams of co2 will be produced 7 g of c3h8 and 98 g of o2
Answers: 1

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 22.06.2019 11:00
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1
You know the right answer?
How many chlorine atoms would be in 6.02 x 10^23 units of gold iii chloride...
Questions

Advanced Placement (AP), 24.07.2021 14:00

Social Studies, 24.07.2021 14:00



Computers and Technology, 24.07.2021 14:00

Geography, 24.07.2021 14:00



Mathematics, 24.07.2021 14:00


Social Studies, 24.07.2021 14:00


Mathematics, 24.07.2021 14:00


Biology, 24.07.2021 14:00

Mathematics, 24.07.2021 14:00




Mathematics, 24.07.2021 14:00

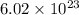 units of gold III chloride is
units of gold III chloride is 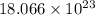
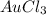
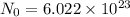
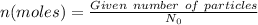
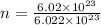
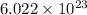 = 1 mole of
= 1 mole of