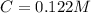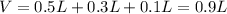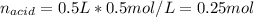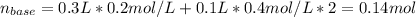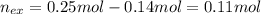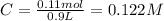Chemistry, 02.01.2020 23:31 PlzNoToxicBan
The following solutions are added together: .5l of .5m hcl, 300 ml of .2m naoh, and 100ml of .4m ca(oh)2. calculate the concentration of the excess acid or base

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution? a. 3.88 m, b. 1.03 m, c. 1.5 m, d. 15.5 m
Answers: 3

Chemistry, 22.06.2019 12:30
A50.0 ml sample of gas at 20.0 atm of pressure is compressed to 40.0 atm of pressure at constant temperature. what is the new volume? 0.0100 ml 0.325 ml 25.0 ml 100. ml
Answers: 1

Chemistry, 22.06.2019 21:00
Rays from the sun are not considered matter true or false
Answers: 2

Chemistry, 23.06.2019 03:30
In chemistry, the type of an atom (what element it is) is determined by: a) the number of protons it contains in its nucleus.b) the number of neutrons it contains in its nucleus.c) the number of protons it has in a cloud around the nucleus.d) the number of neutrons it has in a cloud around the nucleus.e) the number of electrons it exchanges with its neighbors.
Answers: 1
You know the right answer?
The following solutions are added together: .5l of .5m hcl, 300 ml of .2m naoh, and 100ml of .4m ca...
Questions

Mathematics, 15.04.2021 16:00

Physics, 15.04.2021 16:00

Mathematics, 15.04.2021 16:00


Mathematics, 15.04.2021 16:00




Mathematics, 15.04.2021 16:10





Computers and Technology, 15.04.2021 16:10

Mathematics, 15.04.2021 16:10

Mathematics, 15.04.2021 16:10

History, 15.04.2021 16:10


History, 15.04.2021 16:10

History, 15.04.2021 16:10