
Chemistry, 03.01.2020 03:31 deanlmartin
According to the equation above, how many moles of potassium chlorate, kclo3, must be decomposed to generate 1.0 l of o2 gas at standard temperature and pressure?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which type of bond is present in hydrogen sulfide (h2s)? the table of electronegativities is given. a. hydrogen b. ionic c. nonpolar covalent d. polar covalent
Answers: 1

Chemistry, 22.06.2019 06:30
Use examples from the article to explain one positive and one negative effect that chemistry has had on society.
Answers: 2

Chemistry, 22.06.2019 07:20
Part b: study of equilibrium on solubility: mg(oh)2(s) ⇌ mg2+(aq) + 2 oh–(aq) cloudy clear (pink) 7. a. b. 8. a. b. 9. 10. 11. 12. when adding concentrated hydrochloric acid, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 7a. you should indicate which ion was added to or removed from the equilibrium mixture. when adding edta, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 8a. you should indicate which ion was added to or removed from the equilibrium mixture. upon heating in which direction is the equilibrium shifting? upon cooling in which direction is the equilibrium shifting? is the forward reaction a. endothermic explain your answers to questions 9, 10, and 11. (l) left (r) right (l) left (r) right b. exothermic
Answers: 1

Chemistry, 22.06.2019 11:00
Which element would mostly likely have an electron affinity measuring closest to zero
Answers: 3
You know the right answer?
According to the equation above, how many moles of potassium chlorate, kclo3, must be decomposed to...
Questions



Mathematics, 20.07.2020 16:01

English, 20.07.2020 16:01

Mathematics, 20.07.2020 16:01



Mathematics, 20.07.2020 16:01

Mathematics, 20.07.2020 16:01

Biology, 20.07.2020 16:01




Mathematics, 20.07.2020 16:01

Business, 20.07.2020 16:01



English, 20.07.2020 16:01


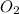 = 1.0 L
= 1.0 L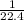 mole
mole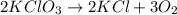
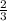 moles of potassium chlorate undergoes reaction.
moles of potassium chlorate undergoes reaction. 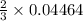 moles of potassium chlorate undergoes reaction.
moles of potassium chlorate undergoes reaction. 


