
Chemistry, 05.10.2019 10:30 sitstareplay7073
Given the following balanced reaction between nitrogen gas and oxygen gas to produce nitrous oxide gas, how many moles of nitrous oxide gas are produced from 1.65 moles of nitrogen gas?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:00
Use examples from the article to explain one positive and one negative effect that chemistry has had on society
Answers: 2

Chemistry, 22.06.2019 01:30
Reaction rate depends on how many molecules are coming into contact with each other with enough energy to react. increasing the temperature of the reactants will increase -
Answers: 3

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 12:20
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l.s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3
You know the right answer?
Given the following balanced reaction between nitrogen gas and oxygen gas to produce nitrous oxide g...
Questions


Mathematics, 12.07.2019 14:30



Spanish, 12.07.2019 14:30






Social Studies, 12.07.2019 14:30

Mathematics, 12.07.2019 14:30

Biology, 12.07.2019 14:30



English, 12.07.2019 14:30

Spanish, 12.07.2019 14:30


English, 12.07.2019 14:30

Spanish, 12.07.2019 14:30

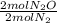 = 1,65 mol N₂O
= 1,65 mol N₂O

