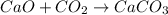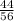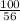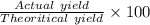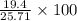Chemistry, 13.01.2020 06:31 jasmin5285
Consider the reaction between calcium oxide and carbon dioxide: cao ( s ) + co 2 ( g ) → caco 3 ( s ) a chemist allows 14.4 g of cao and 13.8 g of co 2 to react. when the reaction is finished, the chemist collects 19.4 g of caco 3 . determine the limiting reactant, theoretical yield, and percent yield for the reaction.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 14:30
100 grams of molten lead (600°c) is used to make musket balls. if the lead shot is allowed to cool to room temperature (21°c), what is the change in entropy (in j/k) of the lead? (for the specific heat of molten and solid lead use 1.29 j/g⋅°c; the latent heat of fusion and the melting point of lead are 2.45 × 104 j/kg and 327°c, respectively.)
Answers: 1

Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3

Chemistry, 23.06.2019 00:30
•hydration •dissociation •dissolving which one goes to which
Answers: 1

Chemistry, 23.06.2019 00:30
Which radioisotope is used to date fossils? a. oxygen-16 b. carbon-14 c. uranium-238 d. carbon-12
Answers: 2
You know the right answer?
Consider the reaction between calcium oxide and carbon dioxide: cao ( s ) + co 2 ( g ) → caco 3 ( s...
Questions

History, 16.10.2020 19:01


Mathematics, 16.10.2020 19:01

Mathematics, 16.10.2020 19:01

Mathematics, 16.10.2020 19:01

Mathematics, 16.10.2020 19:01

Mathematics, 16.10.2020 19:01

Mathematics, 16.10.2020 19:01

Mathematics, 16.10.2020 19:01

Mathematics, 16.10.2020 19:01


Mathematics, 16.10.2020 19:01

History, 16.10.2020 19:01


History, 16.10.2020 19:01

Mathematics, 16.10.2020 19:01




Advanced Placement (AP), 16.10.2020 19:01