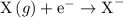Chemistry, 07.01.2020 03:31 Manuelperez1373
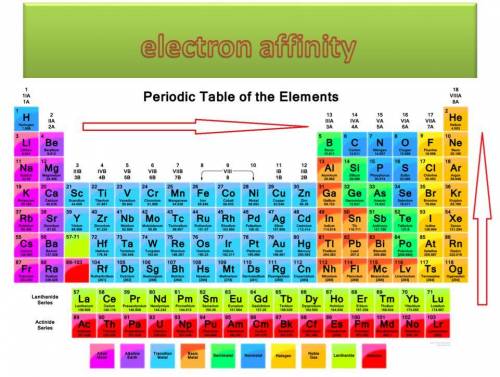
The most negative electron affinity is most likely associated with which type of atoms?
large nonmetal atoms
small nonmetal atoms
large metal atoms
small metal atoms

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Why is permeable soil best for plants that need a lot of drainage?
Answers: 1

Chemistry, 22.06.2019 09:20
Explain that newton first law,second law and third law of motion?
Answers: 2


Chemistry, 23.06.2019 02:30
What role does weathering have in shaping earth’s surface? a) it allows sediments to fall out of a medium. b) it sediments settle on a new surface. c) it breaks down older material into sediments. d) it transports sediments to a different location. will give brainliest, answer quickly.
Answers: 2
You know the right answer?
The most negative electron affinity is most likely associated with which type of atoms?
...
...
Questions






Social Studies, 01.04.2021 16:40



Arts, 01.04.2021 16:40



Mathematics, 01.04.2021 16:40


Mathematics, 01.04.2021 16:40




Mathematics, 01.04.2021 16:40


English, 01.04.2021 16:40