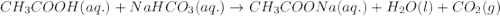Chemistry, 16.01.2020 00:31 gabriella80
Question 5 (essay worth 4 points)
(01.05 mc)
during an experiment vinegar was added to baking soda. the following observations were recorded.
observation 1: the container with the reaction mixture feels cooler.
observation 2: gas bubbles are released.
based on the observation, justify the type of change (physical or chemical) that took place.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Which solution is an example of a nonelectrolyte? a. iodine in hexane b. sodium nitrate in waterc. acetic acid in waterd. hydrogen chloride in water
Answers: 2

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2


Chemistry, 23.06.2019 02:00
As light moves from one material into the next, which of the following affects how much the light waves will refract, or bend? angle at which the ray strikes the medium color of the material density of the material temperature of the light wave
Answers: 2
You know the right answer?
Question 5 (essay worth 4 points)
(01.05 mc)
during an experiment vinegar was added to b...
(01.05 mc)
during an experiment vinegar was added to b...
Questions



English, 22.08.2020 19:01

Biology, 22.08.2020 19:01

History, 22.08.2020 19:01

Engineering, 22.08.2020 19:01




Mathematics, 22.08.2020 19:01







Computers and Technology, 22.08.2020 19:01