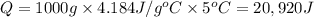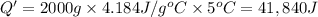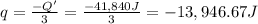Chemistry, 17.01.2020 06:31 dbn4everloved
A1.0 g sample of a cashew was burned in a calorimeter containing 1000. g of water, and the temperature of the water changed from 20.0°c to 25.0°c. in another experiment, a 3.0 g sample of a marshmallow was burned in a calorimeter containing 2000. g of water, and the temperature of the water changed from 25.0°c to 30.0°c. based on the data, which of the following can be concluded about the energy content for 1.0 g of each of the two substances? (the specific heat of water is 4.2 j/(g⋅° (a) the combustion of 1.0 g of cashew releases less energy than the combustion of 1.0 g of marshmallow. (b) the combustion of 1.0 g of cashew releases the same amount of energy as the^combustion of 1.0 g of marshmallow. (c) the combustion of 1.0 g of cashew releases more energy than the combustion of 1.0g of marshmallow. (d) no comparison can be made because the two systems started with different masses of food, different masses of water, and different initial temperatures.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:30
Select each correct answer. more than one answer may be correct. which of the following is a characteristic of unicellular organisms? they can possess tissues and organs. all of their functions are performed by a single cell. they are usually microscopic. each of their cells is specialized to perform a specific function.
Answers: 1

Chemistry, 22.06.2019 07:20
After watching the video "zinc strip in copper nitrate solution", and reading the instructions, click on the link labeled "start" just below the drawing of the pencil tip. follow the direction to complete the 3x3 grid. answer the below questions for the portion of the activity in which sn(s) is placed in agno3(aq)
Answers: 1


Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2
You know the right answer?
A1.0 g sample of a cashew was burned in a calorimeter containing 1000. g of water, and the temperatu...
Questions


English, 29.11.2021 22:20

Chemistry, 29.11.2021 22:20

Biology, 29.11.2021 22:20



Business, 29.11.2021 22:20





Mathematics, 29.11.2021 22:20

History, 29.11.2021 22:20


Computers and Technology, 29.11.2021 22:20

English, 29.11.2021 22:20



Mathematics, 29.11.2021 22:20

Mathematics, 29.11.2021 22:20