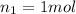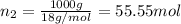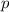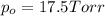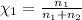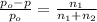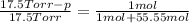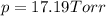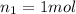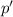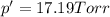Chemistry, 18.01.2020 00:31 naomicervero
Does adding 1 mol of nacl to 1 kg of water lower the vapor pressure of water to the same extent, a lesser extent, or a greater extent than adding 1 mol of c6h12o6 to 1 kg of water? and why?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 16:00
Measuring which physical property is most likely to produce the most precise results when trying to identify a substance
Answers: 1

Chemistry, 22.06.2019 04:00
What layer of the atmosphere is directly above the troposphere?
Answers: 1

Chemistry, 22.06.2019 21:40
Tooth enamel consists mainly of the mineral calcium hydroxyapatite, ca_10(po_4)_6(oh)_2. trace elements in teeth of archaeological specimens provide anthropologist with clues about diet and diseases of ancient people. students at hamline university measured strontium in enamel from extracted wisdom teeth by atomic absorption spectroscopy. solutions with a constant total volume of 10.0 ml contained 0.726 mg of dissolved tooth enamel plus variable concentrations of added sr. added sr find the concentration of sr in the 10 ml sample solution in parts per billion = ng/ml. find the concentration of sr in tooth enamel in parts per million = mu g/g.
Answers: 2

Chemistry, 22.06.2019 21:50
28. which is not a reason that water is used to store spent fuel rods from nuclear power plants? water increases the speed of the chain reaction in the fuel rods. water protects nuclear power plant workers from the high temperature and radiation of the fuel rods. water acts as a radiation shield to reduce the radiation levels. water cools the spent rods. salts action
Answers: 1
You know the right answer?
Does adding 1 mol of nacl to 1 kg of water lower the vapor pressure of water to the same extent, a l...
Questions


Mathematics, 24.05.2021 16:30


Mathematics, 24.05.2021 16:30


English, 24.05.2021 16:30

Mathematics, 24.05.2021 16:30


Chemistry, 24.05.2021 16:30


Social Studies, 24.05.2021 16:30

Mathematics, 24.05.2021 16:30

Mathematics, 24.05.2021 16:30




English, 24.05.2021 16:30

Mathematics, 24.05.2021 16:30

Biology, 24.05.2021 16:30

Mathematics, 24.05.2021 16:30

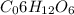 to 1 kg of water.
to 1 kg of water.