Fe(s) + 2hcl(aq) --> fecl2(aq) + h2(g)
when a student adds 30.0 ml of 1.00 m hcl to...

Chemistry, 23.01.2020 03:31 coollid876
Fe(s) + 2hcl(aq) --> fecl2(aq) + h2(g)
when a student adds 30.0 ml of 1.00 m hcl to 0.56 g of powdered fe, a reaction occurs according to the equation above. when the reaction is complete at 273 k and 1.0 atm, which of the following is true?
a) hcl is in excess, and 0.100 mol of hcl remains unreacted.
d) 0.22 l of h2 has been produced.
the correct answer is d. i can't figure out why a is wrong.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Aside from human impact, which of the following causes less water vapor production over a small area? (2 pderivartin
Answers: 1

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3


Chemistry, 23.06.2019 02:00
What causes the appearance of lines in a emission spectrum
Answers: 1
You know the right answer?
Questions


Medicine, 22.06.2020 20:57

Biology, 22.06.2020 20:57

Chemistry, 22.06.2020 20:57




Biology, 22.06.2020 20:57


Mathematics, 22.06.2020 20:57



Mathematics, 22.06.2020 20:57



English, 22.06.2020 20:57

Mathematics, 22.06.2020 20:57

Computers and Technology, 22.06.2020 20:57


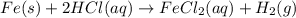
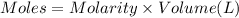
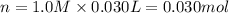
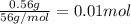
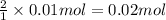 of HCl
of HCl


