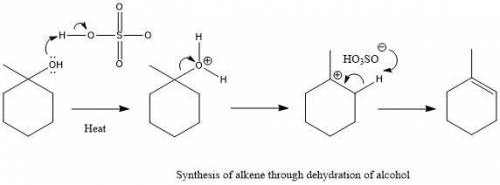
Chemistry, 23.01.2020 07:31 allieallie
In the presence of sulfuric acid, this alcohol is dehydrated to form an alkene through an e1 mechanism. in the box, draw the major alkene product of this reaction.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Which of the following statements is true? a. elements in the last period are radioactive. b. atomic weight is the same as atomic mass. c. elements in the same group have the same number of electron shells. d. atomic number equals the number of neutrons in the nucleus of an atom.
Answers: 1

Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 06:00
This flow chart shows the amount of energy that is emitted by each type of light. ultraviolet > blue light > yellow light > red light (maximum energy) (minimum energy) in an experiment, shining which type of light on a strip of metal would be least likely to produce the photoelectric effect? ultraviolet light dim blue light bright red light bright yellow light
Answers: 2

Chemistry, 22.06.2019 09:00
Chemical energy is a form of a. kinetic energy only. b. both potential and kinetic energy. c. neither potential nor kinetic energy. d. potential energy only. reset
Answers: 1
You know the right answer?
In the presence of sulfuric acid, this alcohol is dehydrated to form an alkene through an e1 mechani...
Questions


English, 22.02.2022 07:10




Mathematics, 22.02.2022 07:10

Mathematics, 22.02.2022 07:10

Mathematics, 22.02.2022 07:10

Computers and Technology, 22.02.2022 07:10




Chemistry, 22.02.2022 07:10





Biology, 22.02.2022 07:10


Chemistry, 22.02.2022 07:10