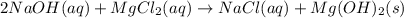Chemistry, 25.01.2020 04:31 Estefaniamarilicolin
Aqueous solutions of sodium hydroxide and magnesium chloride are mixed, resulting in the precipitate formation of magnesium hydroxide with aqueous sodium chloride as the other product. write the balance chemical equation for this reaction

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1

Chemistry, 22.06.2019 05:30
Match the following vocabulary terms to their definitions. 1. amount of energy required to change 1 gram of material from the solid to the liquid state at its melting point 2. a measure of the kinetic energy of the particles of a substance 3. the amount of heat energy required to raise the temperature of 1 gram of liquid water from 14.5°c to 15.5°c 4. amount of energy required to change 1 gram of material from the liquid to the gaseous state at its boiling point 5. the amount of energy required to change 1 gram of a substance 1°c a. temperature b. latent heat of vaporization c. latent heat of fusion d. calorie e. specific heat
Answers: 1

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

You know the right answer?
Aqueous solutions of sodium hydroxide and magnesium chloride are mixed, resulting in the precipitate...
Questions




Business, 05.09.2019 17:30


English, 05.09.2019 17:30


Mathematics, 05.09.2019 17:30


English, 05.09.2019 17:30


English, 05.09.2019 17:30


Mathematics, 05.09.2019 17:30


Mathematics, 05.09.2019 17:30



Mathematics, 05.09.2019 17:30