

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3

Chemistry, 22.06.2019 02:30
You have a sample of a gas that occupies a volume of 17ml at -111 degrees celsius. what volume does the sample occupy at 88 degrees celsius? show all work asap
Answers: 3

Chemistry, 22.06.2019 05:30
According to periodic trend, which of the following most likely has the highest ionization energy? kr be ni sc
Answers: 3

You know the right answer?
At what substrate concentration would an enzyme with a k_cat of 30.0 s⁻¹ and a km of 0.0050 m operat...
Questions




History, 28.06.2019 11:00

English, 28.06.2019 11:00

History, 28.06.2019 11:00


Computers and Technology, 28.06.2019 11:00

Mathematics, 28.06.2019 11:00




Health, 28.06.2019 11:00


Mathematics, 28.06.2019 11:00




Computers and Technology, 28.06.2019 11:00

![v=V_{max}\times \frac{[S]}{(K_m+[S])}=k_{cat}[E_o]\times \frac{[S]}{(K_m+[S])}](/tpl/images/0480/3100/d521f.png)
![V_{max}=k_{cat}[E_o]](/tpl/images/0480/3100/617d3.png)
![[K_m]](/tpl/images/0480/3100/99bb2.png) = Michaelis constant
= Michaelis constant 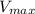 = Maximum rate achieved
= Maximum rate achieved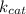 = Catalytic rate of the system
= Catalytic rate of the system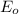 = initial concentration of enzyme
= initial concentration of enzyme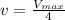
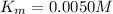
![v=V_{max}\times \frac{[S]}{(K_m+[S])}](/tpl/images/0480/3100/6fc0b.png)
![\frac{V_{max}}{4}=V_{max}\times \frac{[S]}{(0.0050 M+[S])}](/tpl/images/0480/3100/40f70.png)
![[S]=\frac{0.005 M}{3}=1.7\times 10^{-3} M](/tpl/images/0480/3100/96501.png)


