ch4 +2o2 --> co2 + 2h2o

Chemistry, 29.01.2020 06:44 hjohnsonn102010
Methane combusts with oxygen through the following reaction:
ch4 +2o2 --> co2 + 2h2o
how many grams of carbon dioxide are produced when 16.0 g of methane and 48.0 g of oxygen gas combust?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3

Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1

Chemistry, 22.06.2019 15:20
Draw any one of the skeletal structures of a 2° alkyl bromide having the molecular formula of c6h13br and two stereogenic centers. indicate chirality by using wedge and hashed wedge notation. lone pairs do not need to be shown.
Answers: 1

Chemistry, 22.06.2019 20:10
Suppose you mix one mole of sulfuric acid (h2so4) with 1 mole of sodium hydroxide(naoh). why does the ph of the solution remain below 7? ( explain so i can get better understanding! )
Answers: 2
You know the right answer?
Methane combusts with oxygen through the following reaction:
ch4 +2o2 --> co2 + 2h2o
ch4 +2o2 --> co2 + 2h2o
Questions







Chemistry, 21.07.2019 14:30

Mathematics, 21.07.2019 14:30




English, 21.07.2019 14:30

Social Studies, 21.07.2019 14:30




Social Studies, 21.07.2019 14:30

Mathematics, 21.07.2019 14:30


History, 21.07.2019 14:30

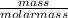
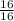 = 1 mole
= 1 mole 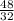 = 1.5 moles
= 1.5 moles 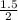 = 0.75mole of carbon dioxide
= 0.75mole of carbon dioxide

