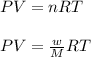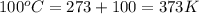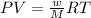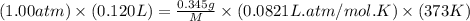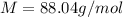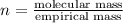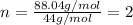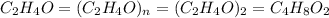Chemistry, 05.02.2020 21:41 2020seogang
Analysis of a volatile liquid showed that it is 54.5% carbon, 9.1% hydrogen, and 36.4% oxygen by mass. a separate 0.345-gram sample of its vapor occupied 120. ml at 100.°c and 1.00 atm. what is the molecular formula for the compound?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 16:00
If 15 drops of ethanol from a medical dropper weight 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? the density of ethanol is 0.80g/ml
Answers: 1


Chemistry, 23.06.2019 03:00
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2
You know the right answer?
Analysis of a volatile liquid showed that it is 54.5% carbon, 9.1% hydrogen, and 36.4% oxygen by mas...
Questions




Mathematics, 17.09.2019 07:20

History, 17.09.2019 07:20





Mathematics, 17.09.2019 07:20

Chemistry, 17.09.2019 07:20



Mathematics, 17.09.2019 07:20





Business, 17.09.2019 07:20

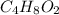
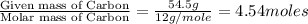
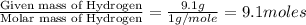
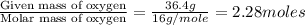
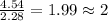
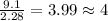
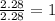
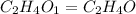
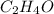 = 2(12) + 4(1) + 16 = 44 g/eq.
= 2(12) + 4(1) + 16 = 44 g/eq.