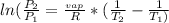Chemistry, 11.02.2020 02:50 angelicar4144
Gasoline is primarily a mixture of hydrocarbons and is sold with an octane rating that is based on a comparison with the combustion properties of isooctane. Gasoline usually contains an isomer of isooctane called tetramethylbutane (C8H18), which has an enthalpy of vaporization of 43.3 kJ/mol and a boiling point of 106.5°C. Determine the vapor pressure of tetramethylbutane on a very hot summer day when the temperature is 38°C.
A. 47.9 torr
B. 67.8 torr
C. 89.3 torr
D. 80.3 torr
E. 36.7 torr
Air (consisting mostly of nitrogen (N2), oxygen (O2), and argon (Ar)) dissolves in water due to?
A. dipole-induced dipole forces.
B. ion–dipole forces.
C. dipole–dipole forces.
D. hydrogen bonding.
E. dispersion forces.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical systems? a) water dissolves nonpolar ionic compounds. b) water dissociates ionic compounds. c) water dissociates covalent molecules. d) water dissolves nonpolar covalent substances.
Answers: 1

Chemistry, 22.06.2019 19:00
Avolleyball player hit a ball with a mass of 0.25 kg. the average acceleration of the ball is 15.5 m/s². how much force did the volleyball player apply to the ball? 62.0 n 3.87 n 62.0 m/s² 3.87 m/s²
Answers: 2

Chemistry, 23.06.2019 01:00
Which polymers are most closely related? a. protein and nucleic acids b. cellulose and starch c. nucleic acids and starch d. nucleic acids and cellulose
Answers: 2

Chemistry, 23.06.2019 11:00
Suppose you increase your walking speed from 7 m/s to 15 m/s in a period of 1 s. what is your acceleration?
Answers: 1
You know the right answer?
Gasoline is primarily a mixture of hydrocarbons and is sold with an octane rating that is based on a...
Questions



History, 05.01.2020 00:31


Spanish, 05.01.2020 00:31

Social Studies, 05.01.2020 00:31

Mathematics, 05.01.2020 00:31


Mathematics, 05.01.2020 00:31

Mathematics, 05.01.2020 00:31


Social Studies, 05.01.2020 00:31


Advanced Placement (AP), 05.01.2020 00:31

Social Studies, 05.01.2020 00:31

English, 05.01.2020 00:31


History, 05.01.2020 00:31