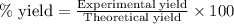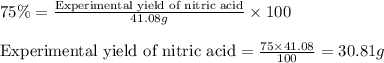Chemistry, 12.02.2020 01:38 jennemylesp19oy5
If the percent yield for the following reaction is 75.0%, and 45.0 g of NO2 are consumed in the reaction, how many grams of nitric acid, HNO3(aq), are produced?
3 NO2(g) + H2O(l) ? 2 HNO3(aq) + NO(g)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:00
What are the first two quantum numbers for the electrons located in subshell 4d? what are the first three quantum numbers for the electrons located in subshell 2s? how many electrons can be held in a sublevel l = 3? how many electrons can be held in the energy level n = 4? how many electrons in an atom can share the quantum numbers n = 4 and l = 3?
Answers: 1

Chemistry, 22.06.2019 21:50
What is a main difference between a mixture and a pure substance? a mixture is only a liquid, but a pure substance can be in any state.a mixture looks the same throughout, but a pure substance does not.1 a mixture can vary in composition, but a pure substance has a set composlo a mixture can be made up of a single compound, but a pure substance car
Answers: 2

Chemistry, 23.06.2019 00:30
Titration reveals that 11.6 ml of 3.0m sulfuric acid are required to neutralize the sodium hydroxide in 25.00ml of naoh solution. what is the molarity of the naoh solution?
Answers: 1

You know the right answer?
If the percent yield for the following reaction is 75.0%, and 45.0 g of NO2 are consumed in the reac...
Questions








English, 19.07.2021 21:10

History, 19.07.2021 21:10



History, 19.07.2021 21:10




Mathematics, 19.07.2021 21:10

Mathematics, 19.07.2021 21:10

Mathematics, 19.07.2021 21:10


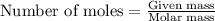 .....(1)
.....(1)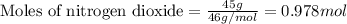
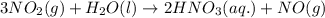
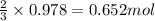 of nitric acid
of nitric acid