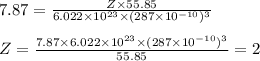Chemistry, 12.02.2020 02:04 chynahbug5676
Metallic iron crystallizes in a cubic lattice. The unit cell edge length is 287 pm. The density of iron is 7.87 g/cm3. How many iron atoms are within a unit cell?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:30
Which of the following would have less momentum than a 52 kg cheetah running at 10 m/s?
Answers: 2

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1

Chemistry, 23.06.2019 02:30
Asubstance is held in an open container. its particles move past one another at random speeds but do not leave the container. heat is removed from the system, and the particles slow down. when enough heat is removed, the particles no longer have enough speed to overcome the weak attractive forces between them. when this happens, the substance enters its solid state. the process described above is known as .
Answers: 3
You know the right answer?
Metallic iron crystallizes in a cubic lattice. The unit cell edge length is 287 pm. The density of i...
Questions




English, 14.07.2020 14:01

Computers and Technology, 14.07.2020 14:01

Mathematics, 14.07.2020 14:01

Mathematics, 14.07.2020 14:01






Computers and Technology, 14.07.2020 14:01

Biology, 14.07.2020 14:01






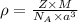
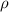 = density =
= density = 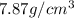
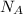 = Avogadro's number =
= Avogadro's number = 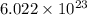
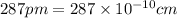 (Conversion factor:
(Conversion factor: 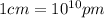 )
)