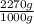Chemistry, 12.02.2020 17:26 deedivinya
3. From the following formula, calculate the quantities required to make 5 lb o hydrophilic ointment. Methylparaben 0.25 g Propylparaben 0.15 g Sodium lauryl sul ate 10 g Propylene glycol 120 g Stearyl alcohol 250 g White petrolatum 250 g Purif ed water, ad 1000 g

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution? a. 3.88 m, b. 1.03 m, c. 1.5 m, d. 15.5 m
Answers: 3

Chemistry, 21.06.2019 23:00
At room temperature what happens to the average kinetic energy of the molecules of a solid, liquid, and a gas
Answers: 2

Chemistry, 22.06.2019 05:30
Arecipe calls for 1.2 cups of oil. how many liters of oil is this?
Answers: 2

Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1
You know the right answer?
3. From the following formula, calculate the quantities required to make 5 lb o hydrophilic ointment...
Questions


Health, 14.12.2020 04:10


Mathematics, 14.12.2020 04:10


Mathematics, 14.12.2020 04:10


History, 14.12.2020 04:10








History, 14.12.2020 04:10



Biology, 14.12.2020 04:10

History, 14.12.2020 04:10