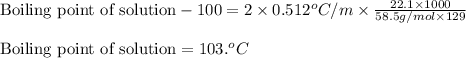Chemistry, 12.02.2020 18:56 stupidjew5496
What are the normal freezing points and boiling points of the following solutions? (a) 22.1 g NaCl in 129 mL of water.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 1

Chemistry, 22.06.2019 15:30
A1.5l container holds p.50 grams of an unknown gas at a pressure of 0.44 atm and a temperature of 50.c what is the molar mass of the unknown gas
Answers: 1

Chemistry, 22.06.2019 18:30
The table lists the lattice energies of some compounds.compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf.the lattice energy increases as the cations get larger, as shown by lif and licl.the lattice energy decreases as cations get smaller, as shown by nacl and naf.the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3

Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 1
You know the right answer?
What are the normal freezing points and boiling points of the following solutions? (a) 22.1 g NaCl i...
Questions



Mathematics, 24.10.2019 20:43




History, 24.10.2019 20:43



Mathematics, 24.10.2019 20:43




Mathematics, 24.10.2019 20:43






Business, 24.10.2019 20:43

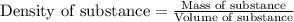
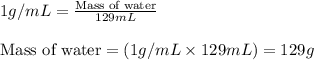
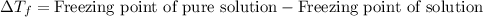
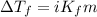
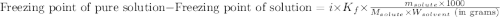
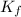 = molal freezing point elevation constant = 1.86°C/m
= molal freezing point elevation constant = 1.86°C/m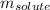 = Given mass of solute (NaCl) = 22.1 g
= Given mass of solute (NaCl) = 22.1 g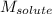 = Molar mass of solute (NaCl) = 58.5 g/mol
= Molar mass of solute (NaCl) = 58.5 g/mol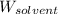 = Mass of solvent (water) = 129 g
= Mass of solvent (water) = 129 g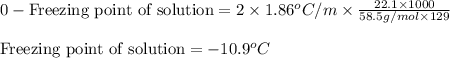
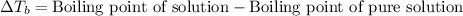
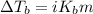
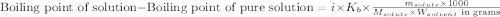
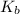 = molal boiling point elevation constant = 0.512°C/m
= molal boiling point elevation constant = 0.512°C/m