

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 13:00
Which of the following are good traits of a hypothesis? it will be able to be testedit can predict an outcomeit will explain the observationsall of these
Answers: 2

Chemistry, 22.06.2019 22:30
Which process describes vaporization that takes place below the surface of a liquid? condensation melting boiling evaporation
Answers: 1

Chemistry, 23.06.2019 01:30
In what way do investigations build scientific knowledge? the results of investigations lead to questions that cannot be tested. they reflect the opinions and social values of scientists, ensuring valid information. the results of investigations lead to new questions, which lead to new investigations. they are not influenced by the research of earlier scientists, so they are able to address gaps in understanding.i
Answers: 1

Chemistry, 23.06.2019 02:00
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1
You know the right answer?
Using the systematic approach for equilibrium problems, calculate the pH of 0.05 M HOCl. Ka= 3.0*10-...
Questions

Chemistry, 09.07.2019 08:30

Mathematics, 09.07.2019 08:30


Mathematics, 09.07.2019 08:30





Mathematics, 09.07.2019 08:30


English, 09.07.2019 08:30


Mathematics, 09.07.2019 08:30

Physics, 09.07.2019 08:30

Social Studies, 09.07.2019 08:30


Mathematics, 09.07.2019 08:30


Mathematics, 09.07.2019 08:30

Biology, 09.07.2019 08:30

![Ka=\frac{[ClO-]*[H+]}{[HClO]}=\frac{x*x}{0.05-x}=3x10^{-8}](/tpl/images/0510/1394/ee229.png)
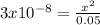
![x=3.87x10^{-5}=[H+]=[ClO-]](/tpl/images/0510/1394/d0c35.png)
![pH=-log[H+]=-log[3.87x10^{-5}]=4.41](/tpl/images/0510/1394/b0e39.png)


