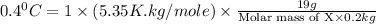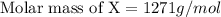Chemistry, 13.02.2020 18:46 kingdrex4772
When 19g of a certain molecular compound X are dissolved in of benzonitrile , the freezing point of the solution is measured to be . Calculate the molar mass of X.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Problem #3 (ch. 1, problem 15)the ideal gas law provides one way to estimate the pressure exerted by a gas on a container. the law isí‘ťí‘ť=푛푛푛푛푛푛푉푉more accurate estimates can be made with the van der waals equationí‘ťí‘ť=푛푛푛푛푛푛푉푉â’푛푛푟푟â’푞푞푛푛2푉푉2where the term nb is a correction for the volume of the molecules and the term an2/v2is a correction for molecular attractions. the values of a and b depend on the type of gas. the gas constant is r, the absolutetemperature is t, the gas volume is v, and the number of moles of gas molecules is indicated by n. if n = 1 mol of an ideal gas were confined to a volume of v = 22.41 l at a temperature of 0â°c (273.2k), it would exert a pressure of 1 atm. in these units, r = 0.0826.for chlorine gas (cl2), a = 6.49 and b = 0.0562. compare the pressure estimates given by the ideal gas law and the van der waals equation for 1 mol of cl2 in 22.41 l at 273.2 k. what is the main cause of the difference in the two pressure estimates, the molecular volume or the molecular attractions?
Answers: 1


Chemistry, 22.06.2019 17:20
Pegmatites are igneous rocks in which the individual minerals are very large. typically, the minerals are all light-colored quartz, feldspar and muscovite. if you were given a black and white photograph of a pegmatite in a quarry (where the rock has been blasted and broken), what physical properties could you use to identify those three minerals in this hypothetical photo? describe each mineral and the specific diagnostic properties. be specific.
Answers: 2

Chemistry, 22.06.2019 20:00
What is the molarity of the solution produced when 145 g of nacl is dissolved in sufficient water to prepare 2.75 l of solution?
Answers: 1
You know the right answer?
When 19g of a certain molecular compound X are dissolved in of benzonitrile , the freezing point of...
Questions

History, 04.02.2021 01:00


Mathematics, 04.02.2021 01:00

History, 04.02.2021 01:00

English, 04.02.2021 01:00

Mathematics, 04.02.2021 01:00


Mathematics, 04.02.2021 01:00


Spanish, 04.02.2021 01:00



Mathematics, 04.02.2021 01:00


Mathematics, 04.02.2021 01:00

Mathematics, 04.02.2021 01:00




History, 04.02.2021 01:00

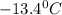 . Calculate the molar mass of X. Freezing point of pure benzonitrile =
. Calculate the molar mass of X. Freezing point of pure benzonitrile = 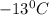
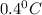
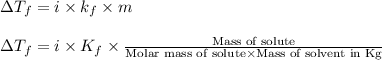
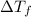 = change in freezing point =
= change in freezing point = 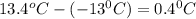
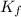 = freezing point constant = 5.35 K.kg/mole
= freezing point constant = 5.35 K.kg/mole